
The last of the giants
Kauri create shelter and nourishment for other species to grow, but now, a disease without a cure is killing these forest giants one by one. In the past five years, the infection rate of kauri has more than doubled in the only forest where it’s monitored—the Waitakere Ranges. At least one in five trees there are doomed. Can we save the species?
The light falls in long heavy bars through the forest. The sun has just risen, and the rickers cast slender shadows across our path. This is a grove of lamp-posts, of ship’s spars, straight and true. On the breeze is the mineral taste of salt from the ocean below.
Fredrik Hjelm and I are walking through Auckland’s lungs, the Waitakere Ranges, a green cloak laid over the length of the city’s west coast. At the edge of the city, the last suburbs melt into the forest, and then there’s nothing but trees, all the way to the cliffs and the sea.
This is where Hjelm feels most at home. He walks through the forest like a docent leading a tour through an art gallery, stopping every few paces to point out details in the corners: native orchids, which grow only in the leaf litter of kauri, young dracophyllum, spiderwebs finely laced in dew.

“This is sinclairii, with the small leaves; it’s one of my absolute favourites. This as well, this is alseuosmia macrophylla; they have really deliciously smelling flowers. They might be a little bit over flowering now. I think it means perfume in Latin. Yeah, smell that.”
This is all regenerating forest—the ancient trees were logged a long time ago—which means the kauri surrounding us are young, their bark still smooth. You could walk along this track without realising anything is the matter. For a long time, people did.
Maybe one clue is the light—there’s too much of it. Ahead of me, Hjelm has stopped to gaze upwards.
The canopy is tattered like old lace. Most of the rickers end in spear-points. Some have stunted branches, like amputated arms. Some still bear leaves, but these are turning copper and yellow. All of them are dead, or nearly so.
Hjelm has seen plenty of dying kauri before, but this ridge above Piha is a particularly bad spot. He points, off the track, to the forms of trees that have rotted and fallen. High winds bring the dead ones down.
Over the last three years, Hjelm walked most of the Waitakere Ranges’ 256 kilometres of track, part of a team which examined 22,744 kauri for signs of infection. He can spot a thinning canopy where most people wouldn’t notice any sign of ill health. The survey found that 80 per cent of kauri here on Maungaroa Ridge are dead or dying; about a quarter of trees in the regional park are infected.
“You’re such a big tree-lover and then you do this,” he says, and shakes his head. “It’s like walking into a hospital, sick trees here and sick trees there.”
This isn’t a hospital, I think to myself. It’s a cemetery.
[Chapter Break]
Kauri are very sensitive to root damage and soil compaction, so losing an individual tree isn’t out of the ordinary. But as the canopy on Maungaroa Ridge turned from green to rust, a handful of people noticed that it wasn’t just one tree, but most of them.
One of those people was a botanical detective. Ross Beever from Landcare Research had just solved the mystery of sudden cabbage-tree death; now he turned his attention to reports of large stands of kauri mysteriously dying. Thirty years earlier, an area of kauri on Great Barrier Island had succumbed en masse, an aberration at the time, but now the same thing was happening in Waipoua, Northland. And here.
This was in 2006. Beever and his colleague, plant pathologist Ian Horner from Plant & Food Research, soon found a suspect in soil samples: a phytophthora.
Phytophthoras are a family of water moulds, which makes them sound sort of funny and harmless, the kind of thing that grows on carrots at the back of the fridge.

Their name tells a different story—phytophthora is Greek for ‘plant-destroyer’. More than a hundred members of the family are busy wreaking havoc on horticultural industries and natural ecosystems around the world. They are fast, invisible, impossible to eradicate. In their home territories, phytophthoras live in balance with the ecosystem in which they evolved. When they travel abroad, that’s when the problems start.
The most famous member of the family, Phytophthora infestans, hitched a ride from Mexico to Europe in the early 1800s. It had a particular taste for potatoes, and from 1845 onwards, it obliterated Ireland’s potato crop, resulting in the deaths of a million people and the emigration of a quarter of the country’s population. (Put it this way: a water mould, too small to be seen, permanently altered the demography and culture of an entire nation.)
New Zealand’s avocado industry is plagued by Phytophthora cinnamomi—one of the most invasive species in the world—and avocado trees are treated with phosphite every year to boost their immunity. Cinnamomi is present in many New Zealand forests, too, and Beever and Horner suspected it was killing kauri.
But after testing soil from Waipoua, they couldn’t prove a connection between the pathogen and the dead trees. Something didn’t add up. On Maungaroa Ridge, Beever and Horner took more samples, and sure enough, these contained a phytophthora. But this time, Horner looked more closely. The phytophthora wasn’t the right shape to be cinnamomi—or any of the others, for that matter. It was a disease unknown to science, and one whose only known victim was kauri.
[Chapter Break]
You can smell a dying tree before you see it: the sweet, sharp scent of resin. Sap bleeds from the trunk, from one place at first, but eventually, the bleeding encircles the tree, preventing water and nutrients travelling to the canopy and upper branches.
The pathogen colonises only the roots.
Below ground, they turn black, and one by one, the cells in the tree’s conductive tissues are killed. Imagine losing your veins. Unable to draw food or water from the earth, the tree starves. There’s no cure.
Once a tree is infected, the only question is how long it will take to die.
In 2014, the new phytophthora received its own name, Phytophthora agathidicida—the second word means ‘kauri-killer’. It’s usually called kauri dieback.
Hjelm and I turn a corner of the track and there’s the source of the fragrance. Bleeding on the ricker ahead is about a metre high—like wax that has melted around the base of a candle, a frozen waterfall of sap. The trees bleed when sap reaches the dead cells, now dead ends, and can’t travel further, bursting out of the trunk instead.
Hjelm reaches up to examine the bleed, and a bracelet of brightly coloured wooden beads peeks out of the cuff of his shirt.

His name and accent are Swedish, but he’s been here for a while now, and he knows the New Zealand forest better than most. He has two young daughters, but apart from them, his life revolves around trees. As well as running his business, The Living Tree Company, he voluntarily leads forest tours for schools and community groups and fellow arborists.
He talks about places which have “really good trees”, and sometimes he adds them to the Notable Trees Register online, where anyone can list a special tree. When he goes on holiday, he looks up trees on the register and visits them. There was an especially good puriri in Opotiki.
“A tree is an asset to a whole community,” he says.
[Chapter Break]
We think of trees as individuals rather than communities. But if a kauri dies, it isn’t simply a matter of subtracting a tree from the landscape—the whole texture of the forest changes.
Kauri create deep layers of acidic leaf litter, determining what else can grow around them. A number of plants are found only, or primarily, in association with kauri, and the largest trees host colonies of epiphytes in their branches: mosses, ferns, orchids, lizards, invertebrates, all living high above the forest floor. At least 100 species live in the crown of Tāne Mahuta in Waipoua Forest.
When kauri disappear, the kauri forest goes, too.
It’s easy to see this contrast in the Waitakeres. Areas that were heavily logged are now covered in low, scrubby, impenetrable bush. Stepping into a grove of mature kauri is like emerging from a tunnel into a cathedral; the roof of the forest soars overhead, and far above, a fretwork of branches interlaces without touching. It’s a series of high vaults, the light filtering in from clerestory windows.
Hjelm has brought me to a grove of kauri that are still healthy, and the sight of them is a relief. I put my hand on the trunk of one and the texture is something between leather and stone.
These trees, centuries old, have reached their full height, so now they’ll grow outwards, ever-thicker. They will probably never reach the girth of our most famous kauri, because agathidicida will reach them first.
The Waitakeres are considered to be ground zero for kauri dieback, but only because this is the only forest that has been surveyed comprehensively. Between 2011 and 2016, the number of known infected trees in the Waitakeres more than doubled, from eight per cent to 19 per cent of kauri. Nearly 60 per cent of large kauri areas are infected. Yet this survey probably underestimates the true infection rate, because only trees that showed symptoms were counted, whereas kauri can carry the pathogen for many years without exhibiting any signs of it. Only lab testing can reveal exactly where agathidicida is in the soil. There’s no data on the spread of dieback in other infected forests—we don’t know exactly where it is.
[Chapter Break]
The reproductive cells of agathidicida, called zoospores, have microscopic tails, and they swim through soggy soil to colonise new trees. But how do they know which way to go?
There are no kauri in Dunedin—their southern limit is a line drawn from Raglan to Tauranga—but there’s plenty of agathidicida, growing on vegetable juice in the lab of University of Otago molecular biologist Monica Gerth. She wants to figure out how to confuse the pathogen’s navigation system, and eventually stop it spreading, in a study supported by the Biological Heritage National Science Challenge.
She studies chemotaxis—how microbes ‘see’ or ‘smell’ what’s around them. Phytophthoras might not have eyes or noses, but something draws them through the soil: chemical cues in the environment.
“Lots of my research is focused around how microbes sense the world, and sense each other, and ways of disrupting it,” she says.
When Gerth heard about kauri dieback, she began wondering: How does agathidicida sense kauri roots? What does it use to navigate through the soil?
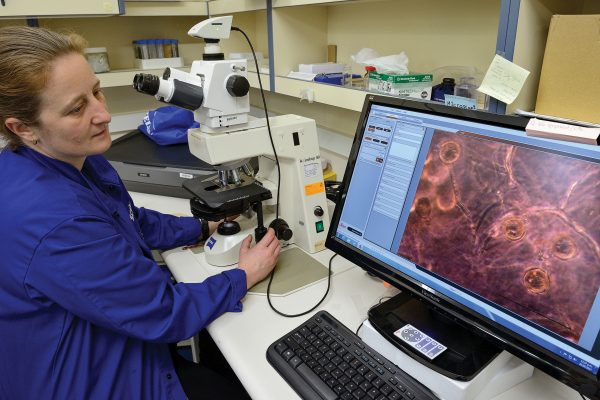
Because agathidicida kills kauri so specifically, she reasoned, there must be something particular about their roots that allows the phytophthora to find it. Kauri is the pathogen’s Southern Cross, its magnetic north, a delicious scent in the breeze. Could we take away its map and replace it with one that is wrong? Give it bad directions? Persuade it to turn back?
Gerth grows agathidicida in her lab on V8 juice solidified with agar. When the pathogen begins to look like a fuzzy mat—a mycelium—she starves it of nutrients and washes it, which tricks it into producing sporangia, bud-like forms that hold zoospores. A cold-shock later and the buds burst open, releasing the zoospores, which are then given a choice of different chemicals in capillary tubes. Gerth is testing root extracts from various native plants to see if they attract or repel zoospores.
“We just watch it in the microscope and see which chemical they pick. Then you count the spores—so say there’s 50 in one tube and none in the other.”
There are thousands of plants Gerth could potentially test for attractiveness or repulsiveness, so rather than screening libraries of chemicals, she began her study by appealing to traditional Māori knowledge of the forest. In collaboration with mātauranga Māori knowledge holders, she’s testing plants that are strongly associated with forest health.
One thing is certain: the zoospores are speedy, in microbe terms. “They’re way faster than bacteria.”
The more we understand agathidicida, the better we’ll be able to fight it—but many features of the pathogen remain mysterious to us.

“I would love to figure out if there’s a particular receptor involved in sensing,” says Gerth. “That’s potentially a target that we could design inhibitors for, or try to block. There’s about 30 of these potential receptors in the phytophthora genome, but which ones they’re actually using for chemotaxis, we have no idea.”
Right now, we don’t know how to kill agathidicida without nuking everything else in the soil, too. The key to getting rid of it would be to target something in its cellular structure that the rest of the ecosystem doesn’t have. (For instance, antibiotics often work by inhibiting enzymes that are involved in the metabolism of one microbe and not another.)
Gerth’s ultimate goal is to design something that would annihilate agathidicida and nothing else—and she’s already found a lead.
“The phytophthora receptors are really weird compared to other receptors,” she says. “They have this extra protein bit dangling off them and we have no idea what that does. So that seems like a really interesting difference that might help us design something specific. It’s outside the scope of this project, but I think it’s very important to explore some of these sorts of fundamental questions about phytophthora biology. It could help us find a cure.”
[Chapter Break]
While Gerth learns more about the pathogen, another group of scientists is plumbing the kauri genome in an attempt to find signs the tree is fighting back.
“Trees have been on this planet a lot longer than we have and they’ve developed a lot of mechanisms for resistance,” says Nari Williams, a plant pathologist at Scion Research. “There have been very few tree diseases where there hasn’t been resistance found.”
Williams is briskly optimistic about the future of kauri. Somewhere within the kauri genome, she says, there is likely to be a form of immunity—it’s just a matter of finding it. She’s leading a six-year research programme looking at how phytophthoras affect several tree species.

For two years, Williams and Stanley Bellgard from Landcare Research collected seeds from significant kauri in forests from Northland to Tauranga. Now, they have 13,000 seedlings growing in their lab, and in another year, they’ll be ready for screening: infecting them with agathidicida to see how they respond. Perhaps one seedling will take a little longer to become infected, or another will appear to be holding out. It might then be bred with another resilient seedling, in an attempt to produce generations of trees that hold out longer, and longer, and longer. The ultimate goal is restoration planting, but it’s not exactly going to be a quick fix: “Time is our tyranny,” she laughs.
While Williams waits for trees to grow, she’s planning to take a closer look at kauri that haven’t shown any signs of disease. There are some living in soil riddled with agathidicida, but which appear to be remaining stalwart. Perhaps the risk of infection is different depending on the environment.
“If we understand the spectrum of susceptibility, it may be that some areas are very vulnerable and therefore need more emphasis for management,” she says.
“I think that everyone’s biggest fear is that we’ll lose kauri as a species or we’ll lose that ecosystem, but if only 10 per cent of a given forest was likely to die, that changes how we might manage it.”
Williams is hopeful because she’s seen how destructive phytophthoras can be, and from her point of view, agathidicida isn’t the worst-case scenario. Her last job involved working on Phytophthora cinnamomi in Western Australia, which is far more destructive.
“In some forest systems there, it’s literally a biological bulldozer. What we see here is not an absolute disease front working through the forest. Not every tree is succumbing at the same rate. You have those situations where you know the pathogen is there and there’s no disease. Personally, I think that’s quite encouraging.”
[Chapter Break]
Doing something about kauri dieback is the responsibility of a partnership between the Ministry for Primary Industries (MPI), the Department of Conservation, Auckland Council, and the regional councils of Northland, Waikato and the Bay of Plenty. It’s called the Kauri Dieback Programme, it’s led by MPI, and one of its primary tasks is to involve everybody in preventing the disease taking hold.
Communities, as much as scientists, are the key to fighting kauri dieback. Agathidicida moves incrementally—most of its spread has been caused by people. It hitchhikes on the shoes of walkers, on the paws of dogs, in the tread of tyres, with seedlings transplanted in forest-regeneration efforts, in chainsaws and spades and harnesses and traps. A speck of dirt the size of a pinhead can carry oospores: sleeper cells which survive for months on end in soil, hibernating until they find the roots of another tree.
Shoe-cleaning stations have been installed around kaurilands, but in the Waitakeres at least, these haven’t halted the spread of the disease—71 per cent of the forest’s kauri-dieback zones are within 50 metres of a track. Nearly half the park’s visitors don’t stop to clean their shoes, and you can hardly blame them: the cleaning stations, placed off to the side of the track, look optional. Many tracks feature ankle-deep mud year-round, which is impossible to dislodge with the equivalent of a dish-brush. (The grove of healthy kauri I visited was at the top of a mud-sodden track that originates in the ranges’ largest dieback-infected zone.)

Yet the Kauri Dieback Programme has been happy to level the blame at ordinary people. In a Radio New Zealand interview, MPI conservation adviser Erik van Eyndhoven cited the public’s unwillingness to “modify their recreational behaviour” as the reason for the disease’s spread.
But do the cleaning stations actually work? No one has studied those installed in the Waitakeres. It’s not that cleaning shoes is pointless—getting rid of dirt, all dirt, is the best way to stop the pathogen from spreading—but many cleaning stations cannot accomplish this. The public do not have the tools to prevent the spread of the disease, even if they wanted to. (Special multi-stage shoe-cleaning stations put in place for a trail-running event, The Hillary, were tested and shown to be 100 per cent effective, but these were temporary.)
“Unfortunately, when it comes to the environmental movement, it’s, ‘We have a nightmare and you’re to blame’, and people switch off with that message,” says University of Auckland social scientist Marie McEntee. “I think people will buy in if they feel their contribution will make a difference and they can see some positive messaging.”
But people don’t feel they can make a difference. A Colmar Brunton poll about kauri dieback found that the majority of people surveyed didn’t feel they could do anything to help.
[sidebar-1]
While most people in kaurilands had heard of kauri dieback, they didn’t know the most important detail about it: that it travels in soil that’s spread. And that’s after 10 years of getting the message across.
“There are some real issues in the way dieback has been communicated to the wider community,” says McEntee.
Questions about what the Kauri Dieback Programme’s communications and engagement budget is being spent on—it was $176,364 in the past financial year—were not answered with specifics. Van Eynhoven cited an e-newsletter (as this magazine went to print, one had been sent in 2017), and a Facebook page, which is updated infrequently. According to documents obtained under the Official Information Act, the last recorded meeting of the communications team took place on December 12, 2016. It is not clear if other meetings have taken place since then, because no minutes exist for them.
Rather, the work of getting the message across to the public has been carried out largely by volunteers.
Vivienne McLean first heard about kauri dieback in 2008. It hadn’t reached the Coromandel yet, but McLean is the sort of person who gets things done, so she set up the Coromandel Kauri Dieback Forum with the idea of keeping the peninsula safe.
But agathidicida was already there. In 2014, it was found in Hukarahi Reserve, then a forest near Whangapoua turned out to be riddled with it. The forum sprang into action, running workshops around the Coromandel for the public, for schools, and for tourism operators. It attended market days, made flyers, organised a hui with Ngāti Maru, surveyed the community on awareness, knowledge of dieback and cleaning-station use, ran advertising and publicity campaigns, and put up billboards about dieback at all the entrances to the peninsula.
“We saw that we were filling a big gap, particularly in the community engagement area,” says McLean, who was formerly a communications professional. “We were trying to do new things to connect. We’d been working closely with Waikato Regional Council and DOC, but neither had plans for community engagement. The forum was undertaking practically all the community engagement on the peninsula.”


McLean discovered that people were happy to volunteer their time—they just didn’t want to sit in planning meetings. If the forum had a coordinator to arrange, marshal and deploy its volunteer army, it would achieve much more. DOC’s community fund got it a part-time coordinator for two years, but that money ran out at the end of 2016. McLean approached MPI for funding to continue the work (until then, the forum had received a total of $220 from the Kauri Dieback Programme).
“I felt we’d been doing the programme’s work for it, and we were making good, responsible use of the money we’d been given so far. They actually used us as a model for talking about what community groups can do.”
The forum was rebuffed in its attempts, and in the end, let its coordinator go, unable to offer her employment beyond December 2016. MPI says it does not fund paid staff for community groups.
“Without financial support volunteer groups can’t sustain a meaningful programme of community engagement on the scale that’s needed long term,” says McLean. “The forum delivered a structured programme of work targeting key audiences amongst locals and visitors.”
McLean has already retired once, and she’s on the cusp of being forced to give up her volunteer work as well. In the meantime, no other community group on the Coromandel or in the Waikato has received financial support to tackle kauri dieback. (Over the past two years, $120,000 in philanthropic funding has been awarded to groups in Auckland and Northland.)
For McLean, however, it’s the little things: the lack of information being shared; the feeling that the effort put in by volunteers was disregarded by the people tasked with managing the disease; most of all, the disconnection.
“I did suggest they should get a bunch of passionate community groups together, ask what they were up to, and swap ideas—that would be a real morale booster, and it wouldn’t cost MPI much more than a plate of muffins and a cup of coffee,” says McLean.
“I don’t think there is a clear strategy for community engagement that embraces kaurilands. We came up with a strategic plan. It’s not just that we were a bunch of do-gooders floating around in the dark.”
[Chapter Break]
On the road that cuts through Waipoua, there’s another one, towering over us. It looks like a giant driftwood sculpture planted in the forest by mistake—trunk and bare branches lifted to the sky, scoured white by waves and time. Except that isn’t what happened. The kauri has shed its bark and leaves, and all that remains is smooth inner wood, pale as bone, still as stone. No sap moves beneath the trunk. It’s the ghost of a tree, standing awkwardly on the side of the road, like a life-size model of a skeleton in the corner of the classroom.
Just around the corner, there’s another, the sharp tips of its branches pronging the sky, and then another. They counted their age in centuries, but it took them less than a decade to die. In kauri time that’s a sudden accident—a heart attack.
Kauri have suffered two great tragedies, and now a third is in progress. The first was logging, when more than 99 per cent of the forest giants were taken. The second was the arrival of agathidicida. The third is our failure to do anything about it.
When the survey results of the Waitakeres were published in an Auckland Council report in December 2016, they startled many—but not the scientists involved.
“I’ve been mapping the disease since 2009,” says Bellgard. “The current report—we predicted those rates back in 2013. After only six years’ data, we’d seen what the rates of spread could be in these kauri-dominated areas.”
Scientists working on kauri dieback, however, are stymied. They still don’t have some of the most fundamental information they need. The basics are missing.
It’s impossible to manage the spread of a disease, for instance, until we know where it is, but survey work outside the Waitakeres has been sporadic. When it comes to forests with significant kauri, such as Tāne Mahuta or Te Matua Ngahere, we don’t know how far the pathogen is from important trees.
“It’s not straightforward to detect,” acknowledges Horner. “We keep finding new sites all the time, but it’s really important that we finish that job. We’ve got a lot of dots on the map but it needs a lot of filling in. There’s a lot of areas where we know a particular forest has got some infection in it but we don’t know how far it’s spread.”

Nor do we know which plants agathidicida can live on. Is kauri its only host? Do infected kauri act as islands which the phytophthora must swim between, or can it live on other species, too? A study by University of Auckland masters student Jessica Ryder indicated that seven native species are potential agathidicida hosts, including tanekaha, rewarewa, mingimingi and dracophyllum, but more research is required to confirm her findings.
“If tanekaha or kauri associates can support it, then that makes it a totally different thing to manage in terms of where we route our tracks,” says Te Roroa ecologist Tom Donovan. “Then we have vectors like pigs. We still don’t have really strong evidence to take to the pig-hunting community to say why we need to control pigs.”
Next on the priority list is latency: the mystery of the lag time between the infection of fine roots and symptoms appearing, when the forest giants start losing their hair. Is agathidicida actually spreading in the Waitakeres, or is it simply manifesting?
Many people interviewed for this story were originally part of a technical advisory group formed by MPI, and they repeatedly laid out their research priorities: surveying, host specificity, oospore deactivation, vectors, latency. The Kauri Dieback Programme last convened the group in 2015. (MPI says the scientists were compromised by conflicts of interest.)
Organising science takes a long time, but MPI’s internal processes delay it further. Research into host specificity and oospore deactivation is slated, but has not started. (Oospore deactivation was identified as a “high-priority research project” by the science team in November 2011.) Survey work is underway, and will be completed in the middle of 2018.
MPI has also commissioned work into the monitoring of forest health, locating iconic trees, mapping all kauri, and figuring out how to survey trees from aircraft. Most promising are the trials it has funded into the use of phosphite, which is providing a stay of execution to some trees.
But it’s the lack of urgency that most frustrates onlookers. The Auckland Council report on the Waitakeres was first circulated in December 2016, and its authors called for the council to investigate closing the 16,000-hectare park.
[sidebar-2]
A year later, dismayed by inaction, West Auckland iwi Te Kawerau a Maki placed a rāhui, or ritual prohibition, on the Waitakeres. It called for Auckland Council and MPI to enforce it.
“I don’t want my mokopuna to read reports on the trees that used to stand,” the iwi’s executive chairman, Te Warena Taua, told an Auckland Council meeting in December 2017. At the meeting, the council voted against enforcing the rāhui, opting instead to close an additional 13 of the park’s 80 high- and medium-risk tracks, and step up work on track improvement and cleaning stations. (On December 8, a further 20 closures were announced.)
“We actually find it pretty insulting that they put in the resolution that they understand and respect the principles of the rāhui, while simultaneously ignoring it and undermining it,” says Te Kawerau a Maki executive manager Edward Ashby. “You can’t have it both ways.”
[Chapter Break]
If money represents priorities, kauri would be ranked ‘fairly unimportant’. Compared to funding made available for other biosecurity threats, kauri comes up short.
MPI was allocated $4.8 million over four years to manage, research and communicate about kauri dieback; the response to the freshwater pest didymo furnished $4.6 million over three years for communication alone. In 2002, the government spent $90 million spraying West Auckland to eradicate the painted apple moth, after a previous $23.3 million programme failed.
The disparity continues in science funding. In 2009, the Ministry of Business, Innovation and Employment (MBIE) put $15.2 million into developing kiwifruit cultivars resistant to the devastating Psa bacterium, on top of $25 million committed by the government to the biosecurity response. By contrast, MPI spends about $800,000 a year on kauri dieback science, while MBIE allocated $10 million to research diseases affecting pine trees, horticultural trees—and kauri.
“You’ve got tens of millions of dollars being thrown at the kiwifruit industry to solve Psa, and then you look at what we’ve been trying to scrape an existence from in the kauri space,” says Horner.
“An iconic tree like kauri—how do you value that? You can’t put a monetary value on something that’s got spiritual significance, emotional significance, ecological significance—and you shouldn’t have to.”
Kauri isn’t an export product, or a food source with the backing of billion-dollar industry. Which begs the question: Who stands up for the trees? In this case, it’s been iwi and community groups that have pushed action forward.
Te Kawerau a Maki will take the battle to government. Forests deserve a dedicated protection plan, says Edward Ashby, like the one being discussed for freshwater.
“We actually need a healthy forest policy,” he says. “Kauri dieback is one thing, but now we’ve got myrtle rust, a number of diseases, and then there’s population pressure. There’s just the assumption that the environment can absorb anything you throw at it. It’s that sort of national-level forest-health strategy that needs to happen. I don’t know why it’s not really been debated or talked about. That’s where to next for us.”
Forest health is not unlike human health, says Ian Horner: “The more resilient you are, you might be able to ward off certain issues. A sick tree is going to be more susceptible to a disease than a healthy tree.”

Ecologists use a triangle-shaped model to show that disease is caused by three factors: not just the pathogen, but its host and environment, too. If the environment favours the pathogen, then the host gets sick. But if the environment favours the host, then the host has the strength to fight off the disease.
And the health of kauri cannot be ascertained by looking at them on their own, say iwi leaders. Researcher Tui Shortland interviewed tohunga and kaumātua throughout Northland for a report on mātauranga, or traditional knowledge, of the kauri forest. She used this to develop a series of measurements that other mana whenua can use to assess whether their forests are healthy. (Her report was completed in 2013: it will be put into use by MPI in early 2018.)
“Scientifically, the Māori approach is a sensible one,” says Horner. “Looking at the whole system rather than one specific little bit makes perfect sense. We tend to try and partition everything into a nice little box, and look at that little box alone.”
Meanwhile, there might be a fourth tragedy waiting in the wings. Early on, Australian plant pathologists warned New Zealand that kauri dieback could behave the same way as jarrah dieback, which they were studying. First, the phytophthora wipes out the jarrah. Once that’s gone, it gets started on everything else.
Is the kauri simply the forest equivalent of the canary in the coal mine?
“What we see in other pathogens elsewhere—it happens with myrtle rust, it happens with cinnamomi—you have your most susceptible tree species get taken out first, and then you get a complete shift in the ecology,” says Nari Williams. “That means the other plants are under more stress and they’re exposed to a higher inoculum rate, and then the next things might go.”
The lack of funding is exacerbated by a lack of coordination. Many people interviewed for this story spoke of difficulty in obtaining information, or research being duplicated across regions. Several kauri dieback researchers did not know about other studies funded by the Kauri Dieback Programme. Several people who had advised the programme said they felt they had been part of a box-ticking exercise.
“It’s all very well to say, ‘Oh yeah, we consulted’,” one person told me. “But there’s no listening to the consultation.”
Decision-making is postponed until more information is known, preventing action.
“We can’t sit around waiting for perfect answers before managing things,” says Williams. “You have to take an adaptive management approach, and there’s a hell of a lot that we can learn from similar situations elsewhere. A piecemeal approach is not going to save the forest. It’s basically: Get on with it.”
[Chapter Break]
Stand in front of Te Matua Ngahere and it’s hard to call it just a tree. A weight settles over you. It has a presence.
The forest giants not only create space for other species to grow, but they determine the climate for a different type of human thinking, too. In a country with no ruins or city foundations or pottery shards to connect us with life a thousand years ago, we depend on kauri to unlock the past. Imagine, says Tom Donovan, all the people who have stood in this place and gazed at this tree. Imagine all the people who might still do so.
Kauri are a map to the scale of things, ejecting us from the urgency of the present moment.
I ask Donovan what Te Matua Ngahere means to him, and his thinking is the same as mine. “It’s a connection with the ancient past, and with the distant future,” he says. “Hopefully.”


















